 
In spectral phenomena such as the Zeeman effect it becomes evident that transitions are not observed between all pairs of energy levels. Selection Rules for Electronic Transitions The different orientations of orbital angular momentum represented by the magnetic quantum number can be visualized in terms of a vector model. It is called the magnetic quantum number because the application of an external magnetic field causes a splitting of spectral lines called the Zeeman effect. 
The direct implication of this quantum number is that the z-component of angular momentum is quantized according to While the azimuthal dependence of the wavefunction only requires the quantum number to be an integer, the coupling to the colatitude equation further constrains that integer to be less than or equal to the orbital quantum number. The expression of the separation constant in terms of this quantum number affects the form of the colatitude equation.įrom the azimuthal equation of the hydrogen Schrodinger equation comes a quantum number with the constraint This is the origin of the magnetic quantum number. The constants above are then required to be of the form Since for all integer values n, these represent the same point in space. 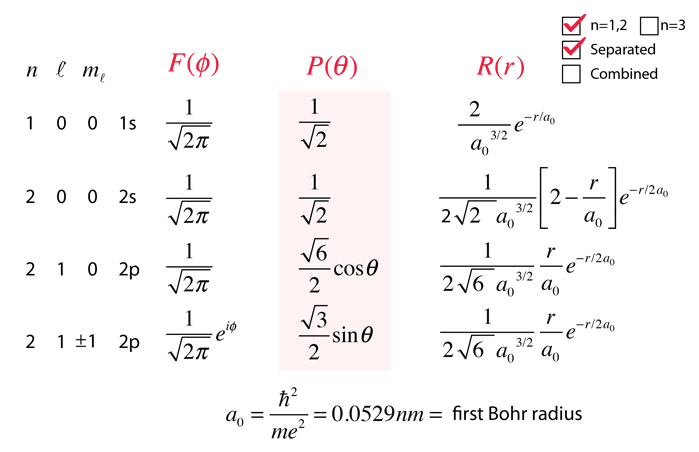
This is the easiest of the three separated equations to solve and gives the formīut physical constraints on the wavefunction require Upon separation of the Schrodinger equation for the hydrogen atom, the azimuthal equation is: Hydrogen Schrodinger Equation The Azimuthal Equation 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
